|

CLICK ON weeks 0 - 40 and follow along every 2 weeks of fetal development
|
||||||||||||||||||||||||||||
How to reverse the age-related loss of stem cells Adult stem cells replenish dying cells and regenerate damaged tissues throughout our lifetime. But, we lose many of those stem cells along with their regenerative capacity as we age. Working in flies and mice, researchers at the Buck Institute for Research on Aging along with other collaborative institutions, discovered that TOR - a nutrient sensing pathway which is central to the aging process - drives the loss of adult stem cells. However, treating mice with TOR-inhibitor rapamycin prevented the loss of stem cells in mouse trachea and could possibly be managed to reverse age-related stem cell loss across species. The research is published in Cell Stem Cell. In most of our tissues, adult stem cells hang out in a quiet state - ready to be activated in case of infection or injury. In response to TOR, stem cells rapidly divide to generate daughter cells that replace damaged tissue. This division has to be "asymmetric," as only one of the two cells generated will differentiate into a tissue cell, the other remains a stem cell. Previous research has shown that TOR maintained at a low level in mice, preserves stem cells in a quiescent state until needed. In this study, researchers closely observed TOR signaling activation of stem cells to regenerate. Buck professor and senior author Heinrich Jasper PhD, now a staff scientist at Genentech, believes TOR activation is not only important for rapid tissue repair, it also increases the probability stem cells, over time, will lose their own ability to differentiate. Such a loss, as observed in the fly intestine, mouse trachea and muscle tissue, is particularly prevalent when tissue is under heavy or chronic pressure to regenerate as in response to infection or severe trauma. "It's all about maintaining a balance between stem cell renewal and differentiation. It's easy to see how a loss of adult stem cells might accrue over a lifetime and accelerate with aging." 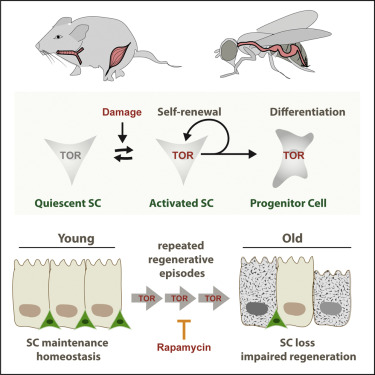 The work at the Buck Institute was led by postdoctoral fellow Samantha Haller PhD, also of Immunology Discovery, at Genentech Inc., whose experiments were on the intestines of fruit flies and later on the mouse trachea, tissues that share many developmental similarities. Experiments involving mouse muscle tissue were done at Stanford University. At the Buck Institute, mice were put on differing regimens of rapamycin that varied by life stage of the mouse. Rapamycin was seen to rescue stem cells even when given to mice at 15 months of age - our human equivalent of age 50. Jasper: "In every case where we saw a decline in the number of stem cells, rapamycin would bring it back." Whether stem cell numbers recovered due to replenishment by differentiated cells, or by an increase in "asymmetric" stem cell divisions, is to be determined. Highlights Somatic stem cells transiently activate mTORC1 signaling during tissue regeneration Chronic mTORC1 activation leads to stem cell loss Repeated regenerative episodes result in mTORC1-dependent loss of SCs Long-term rapamycin exposure limits age-related SC loss in the tracheal epithelium Summary The balance between self-renewal and differentiation ensures long-term maintenance of stem cell (SC) pools in regenerating epithelial tissues. This balance is challenged during periods of high regenerative pressure and is often compromised in aged animals. Here, we show that target of rapamycin (TOR) signaling is a key regulator of SC loss during repeated regenerative episodes. In response to regenerative stimuli, SCs in the intestinal epithelium of the fly and in the tracheal epithelium of mice exhibit transient activation of TOR signaling. Although this activation is required for SCs to rapidly proliferate in response to damage, repeated rounds of damage lead to SC loss. Consistently, age-related SC loss in the mouse trachea and in muscle can be prevented by pharmacologic or genetic inhibition, respectively, of mammalian target of rapamycin complex 1 (mTORC1) signaling. These findings highlight an evolutionarily conserved role of TOR signaling in SC function and identify repeated rounds of mTORC1 activation as a driver of age-related SC decline. Authors: Samantha Haller, Subir Kapuria, Rebeccah R. Riley, Monique N. OLeary, Katherine H. Schreiber, Julie K. Andersen, Simon Melov, Jianwen Que, Thomas A. Rando, Jason Rock, Brian K. Kennedy, Joseph T. Rodgers, Heinrich Jasper Other Buck researchers involved in the study include Samantha Haller, Subir Kapuria, Rebeccah R. Riley, Monique N. O'Leary, Katherine H. Schreiber, Julie K. Andersen, Simon Melov and Brian Kennedy. Other collaborators include Thomas A. Rando and Joseph T. Rodgers, Paul Glenn Laboratories for the Biology of Aging, Stanford University, Stanford, CA; Jianwen Que, Department of Medicine, Columbia University, New York, NY; and Jason Rock, Department of Anatomy, USCF School of Medicine, San Francisco, CA. Citation: mTORC1 activation during repeated regeneration impairs somatic stem cell maintenance DOI: 10.1016/j.stem.2017.11.008 The work was supported by grants from the National Institutes of Health R01 AG041764, R01 AG047497, R01 DK100342. About the Buck Institute for Research on Aging The Buck Institute is the U.S.'s first independent research organization devoted to Geroscience - focused on the connection between normal aging and chronic disease. Based in Novato, California, the Buck is dedicated to extending "healthspan," the healthy years of human life, and does so by utilizing a unique interdisciplinary approach involving laboratories studying the mechanisms of aging and others focused on specific diseases. Buck scientists strive to discover new ways of detecting, preventing and treating age-related diseases such as Alzheimer's and Parkinson's, cancer, cardiovascular disease, macular degeneration, osteoporosis, diabetes and stroke. In their collaborative research, they are supported by the most recent developments in genomics, proteomics, bioinformatics and stem cell technologies. For more information: http://www.thebuck.org. Return to top of page |
Dec 6, 2017 Fetal Timeline Maternal Timeline News News Archive  Top image: single tracheal epithelium after one round of regeneration; Lower image: single tracheal epithelium after 3 rounds of regeneration. GREEN: stem cells. LIGHT BLUE: cell nuclei. Image credit: Samantha Haller PhD.
|
||||||||||||||||||||||||||||

