|

CLICK ON weeks 0 - 40 and follow along every 2 weeks of fetal development
|
||||||||||||||||||||||||||||
|
Developmental biology - Mitochondria Mighty mitochondria flex their DNA power "We haven't been looking at the full complexity of the cellular network. If we're fighting cancer, for example, in only half of our genome, then it's half of a solution. Now we can fight these diseases knowing all their genetic components." The field of intracellular communication has just emerged and accelerated in the last decade. As medical equipment becomes more incisive, scientists can now better detect smaller and smaller molecular mechanisms inside a cell. USC researchers focused on the two parts of the cell that carry DNA: (1) the nucleus and (2) the mitochondria. Most genetic material resides in the nucleus, the largest component of the cell. Nuclear DNA relays 'templates' instructing other cell mechanisms what to do in response to specific stimuli. Within this scheme, the small mitochondria - which carry only maternal genes - function as energy-producing factories, turning molecules from food into fuel to power the entire cell. However, despite being small in size, mitochondria apparently do not just respond to instructions from nuclear DNA. Working with human cells, USC scientists observered that mitochondrial DNA in a cell under stress and starved of nutrients emits a small peptide which consists of amino acids, MOTS-c. This peptide enters the nucleus activating an antioxidant or defensive response. "Most diseases are due to aging, and aging leads to a breakdown in cell functions," explains Lee. "When things go wrong in the body, it's because some cellular mechanism in the body went wrong. So, understanding how cells work means we have more insight into how damage occurs and how we can prevent or fix it." Highlights MOTS-c, a peptide encoded by the mitochondrial DNA, can localize to the nucleus Metabolic stress triggers AMPK-dependent translocation of MOTS-c to the nucleus MOTS-c can bind to chromatin and regulate nuclear gene expression MOTS-c can promote cellular stress resistance against metabolic stress Summary Cellular homeostasis is coordinated through communication between mitochondria and the nucleus, organelles that each possess their own genomes. Whereas the mitochondrial genome is regulated by factors encoded in the nucleus, the nuclear genome is currently not known to be actively controlled by factors encoded in the mitochondrial DNA. Here, we show that MOTS-c, a peptide encoded in the mitochondrial genome, translocates to the nucleus and regulates nuclear gene expression following metabolic stress in a 5'-adenosine monophosphate-activated protein kinase (AMPK)-dependent manner. In the nucleus, MOTS-c regulated a broad range of genes in response to glucose restriction, including those with antioxidant response elements (ARE), and interacted with ARE-regulating stress-responsive transcription factors, such as nuclear factor erythroid 2-related factor 2 (NFE2L2/NRF2). Our findings indicate that the mitochondrial and nuclear genomes co-evolved to independently encode for factors to cross-regulate each other, suggesting that mitonuclear communication is genetically integrated. Authors: Kyung Hwa Kim, Jyung Mean Son, Bérénice A. Benayoun and Changhan Lee. Acknowledgements The study authors include lead researchers Lee, Kyung Hwa Kim, Jyung Mean Son and Berenice A. Benayoun of the USC Leonard Davis School, USC Norris Comprehensive Cancer Center and USC Stem Cell Initiative. Lee is a consultant for, and a shareholder of, CohBar Inc., which is based in Menlo Park, Calif.; the company conducts research and development of mitochondria-based therapeutics. The work was funded by grants from the National Institutes of Health (R01AG052558), the Ellison Medical Foundation, the American Federation for Aging Research and the Hanson-Thorell family to Changhan Lee and National Institutes of Health grant R00AG049934 to B. Benayoun. Return to top of page | Jul 12, 2018 Fetal Timeline Maternal Timeline News News Archive 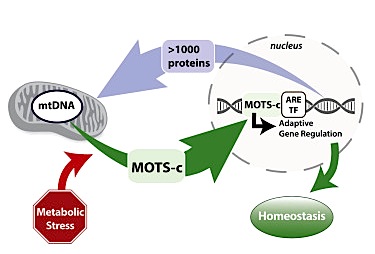 Mitochondria respond to cell stress such as lack of nutrients (starvation) by sending MOTS-c, a peptide, into the nucleus triggering an antioxidant response. Image credit: Lee Lab, University of Southern California.
|
||||||||||||||||||||||||||||

