|
|
Developmental Biology - Jumping Genes
Jumping Genes Help Make Neurons in a Petri Dish
Finding could lead to safer and more effective regenerative therapies for Parkinson's and other brain conditions...
The process of making functional brain cells in a lab dish requires precise activation of selfish genetic elements known as LINE-1 (L1) retro-transposons. This finding by researchers at King Abdullah University of Science & Technology (KAUST), could lead to safer and more effective regenerative therapies for Parkinson's disease and other brain conditions.
The genomes of humans, mice and other mammals have hundreds of thousands of L1 elements. Most are inactive, but some retain the ability to make copies of themselves and jump into different segments of DNA, impacting gene regulation in both harmful and beneficial ways. Sometimes, these jumping genes can trigger disease. In early brain development, however, L1 activity is needed for neurons to form properly although why has never been clear.
To answer this question, Valerio Orlando and colleagues turned to a cellular model of neuronal development.
Working with scientists in Italy, they engineered skin cells taken from mouse embryos to make various "reprogramming factors'". One result was the conversion of skin cells into dopamine producing neurons similar to those found in the substantia nigra located deep in the brain. Through this process, they observed L1 become active.
The cells were treated with two kinds of drugs to block L1 dynamics. Both treatments dramatically impaired cell conversion, demonstrating that "activation is required for successful reprogramming of skin cells into neuronal cells," explains Francesco Della Valle, a postdoc in Orlando's lab group, and the first author of the new study published in Stem Cell Reports.
Researchers then sequenced all the DNA inside the mouse skin cells, both before and after their conversion, to determine where the L1 elements had newly inserted themselves into the genome sequence. They found insertional hotspots around the genes involved in neuronal lineage commitment and neuron function. Consequently, they found DNA at these two sites was less densely packaged, allowing for higher levels of relevant gene expression (function).
"Our work boosts the concept that repetitive elements play an important, unprecedented role in cell differentiation and tissue specific developmental programs," explains Della Valle.
Those insights could prove invaluable as researchers design new kinds of cell therapies to replace the dopamine-producing neurons lost in people with Parkinson's disease and related disorders.
"Aberrant L1 activity could threaten the viability or safety of any such product, while optimizing L1 function could enhance the manufacturing and consistency of this type of regenerative treatment."
Francesco Della Valle PhD, King Abdullah University of Science and Technology (KAUST), Biological Environmental Science and Engineering Division, KAUST Environmental Epigenetics Program, King Abdullah University of Science and Technology, Thuwal, Saudi Arabia
Abstract Highlights
L1 activation accompanies induced dopaminergic neuron maturation
L1 inhibition impairs the transdifferentiation potential of MEFs
L1 retrotransposition creates a lineage-specific genetic mosaicism
L1 insertions correlates with open chromatin and lncRNA transcription
Summary
In mammals, LINE-1 (L1) retrotransposons constitute between 15% and 20% of the genome. Although only a few copies have retained the ability to retrotranspose, evidence in brain and differentiating pluripotent cells indicates that L1 retrotransposition occurs and creates mosaics in normal somatic tissues. The function of de novo insertions remains to be understood. The transdifferentiation of mouse embryonic fibroblasts to dopaminergic neuronal fate provides a suitable model for studying L1 dynamics in a defined genomic and unaltered epigenomic background. We found that L1 elements are specifically re-expressed and mobilized during the initial stages of reprogramming and that their insertions into specific acceptor loci coincides with higher chromatin accessibility and creation of new transcribed units. Those events accompany the maturation of neuronal committed cells. We conclude that L1 retrotransposition is a non-random process correlating with chromatin opening and lncRNA production that accompanies direct somatic cell reprogramming.
Authors
Francesco Della Valle, Manjula P. Thimma, Massimiliano Caiazzo, Salvatore Pulcrano, Mirko Celii, Sabir A. Adroub, Peng Liu, Gregorio Alanis-Lobato, Vania Broccoli and Valerio Orlando.
Acknowledgements
The authors are grateful to Fred Gage (Salk Institute) for the L1-EGFP Plasmid and stimulating discussions. John Goodier for pGF21 and pTN201 plasmids. Edith Heard for pTNC7 plasmid. V.O. is supported by King Abdullah University of Science and Technology ( BAS 1037/01-01 , CRG URF 126310101). M.C. is supported by Italian Ministry of University and Education grant MIUR FIR 2013 RBFR13LH4X_002 . V.B. is supported by Italian Ministry of University and Education , Italian Institute of Technology Seed project, Telethon Foundation , Cariplo Foundation , and Michael Fox Foundation and ERC grant AdERC #340527 . Thanks to Dr. Giancarlo Bellenchi for sharing reagents and laboratory facilities.
Return to top of page.
| |
|
Apr 15 2020 Fetal Timeline Maternal Timeline News
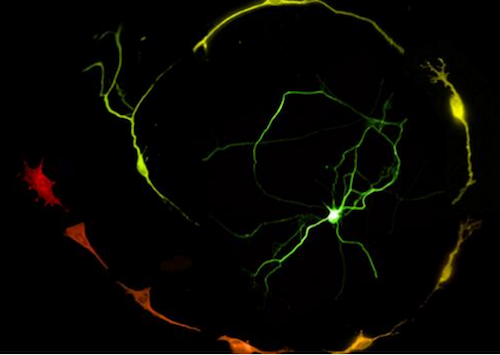
Engineered Skin cells (RED) become dopanine-producing Neurons (GREEN).
CREDIT 2020 Della Valle et al.
|



